Bisphenol
The bisphenols (/ˈbɪsfɪnɒl/) are a group of industrial chemical compounds related to diphenylmethane; commonly used in the production of plastics and epoxy resins. They feature two hydroxyphenyl groups linked by a methylene bridge. Also included are bisphenol S, P, and M. "Bisphenol" is a common name; the letter following denotes the variant, which depends on the additional substituents. Bisphenol A is the most common representative of the group, with millions of metric tons produced globally in the past decade, often simply called "bisphenol".[1]
Bisphenols have attracted controversy because of their role as endocrine disruptors. Their estrogenic properties are weak, but the scale of their production is considerable.[2][3]
Partial inventory of bisphenols
[edit]| Structural formula | Name | CAS | Reactants | |
|---|---|---|---|---|
| Bisphenol A | 80-05-7 | Phenol | Acetone | |
 |
Bisphenol AP | 1571-75-1 | Phenol | Acetophenone |
 |
Bisphenol AF | 1478-61-1 | Phenol | Hexafluoroacetone |
 |
Bisphenol B | 77-40-7 | Phenol | Butanone |
 |
Bisphenol BP | 1844-01-5 | Phenol | Benzophenone |
 |
Bisphenol C | 79-97-0 | o-cresol | Acetone |
 |
Bisphenol C 2 | 14868-03-2 | Phenol | Chloral |
| Bisphenol E | 2081-08-5 | Phenol | Ethanal | |
| Bisphenol F | 620-92-8 | Phenol | Formaldehyde | |
 |
Bisphenol G | 127-54-8 | 2-Isopropylphenol | Acetone |
| Bisphenol M | 13595-25-0 | |||
 |
Bisphenol S | 80-09-1 | Phenol | Sulfur trioxide |
 |
Bisphenol P | 2167-51-3 | ||
 |
Bisphenol PH | 24038-68-4 | 2-Phenylphenol | Acetone |
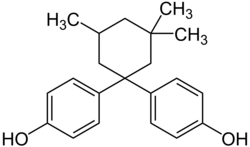 |
Bisphenol TMC | 129188-99-4 | Phenol | 3,3,5-Trimethylcyclohexanone |
 |
Bisphenol Z | 843-55-0 | Phenol | Cyclohexanone |
 |
Dinitrobisphenol A | 5329-21-5 | Bisphenol A | Nitric acid |
 |
Tetrabromobisphenol A | 79-94-7 | Bisphenol A | Bromine |
 |
Tetramethyl bisphenol F | 5384-21-4 | 2,6-Xylenol | Formaldehyde |
Health effects
[edit]Bisphenols A (BPA), F (BPF) and S (BPS) have been shown to be endocrine disruptors, potentially relating to adverse health effects.[2][4] Other implicated health effects include obesity and metabolic disorders.[5]The threat is of interest because the chemical is pervasive in everyday life, inclluding food[2] and thermal paper.[6]
Occurrence in humans
[edit]Bisphenols have monitored in humans through analysis of serum and urine.[7] [8] Workers who manufacture BPA-based thermal paper have high BPA levels.[6]. BPS and BPF levels are elevated among workers dismantling e-waste in China.[9]
Many analogues of BPA, such as bisphenol F (BPF), bisphenol S (BPS), bisphenol AP (BPAP), bisphenol AF (BPAF), bisphenol FL (BPFL), and bisphenol C (BPC) are less studied with respect to their hormone-disrupting potential and other health effects.[10][5][11][12]
Biodegradation
[edit]Due to its high production volumes, BPA has been characterised as "pseudo-persistent",[13] leading to its spreading and potential accumulation in a variety of environmental matrices. BPA has a fairly short half-life. A number of aerobic organisms degrade BPA.[14][15]
Precautions
[edit]Engineering controls have been applied such as process enclosure, local exhaust ventilation, and isolation of bisphenol-handling areas lower airborne and surface contamination in workplaces where bisphenols are used.[16][17].
Exposure to Bisphenol
[edit]Because bisphenol are largely used in everyday life such as in plastics, protective coating of packages, inner lining of food containers, and thermal paper of receipts, it can enter the body through multiple pathways.[18]
Exposure Pathways
[edit]Dietary Exposure
[edit]The main pathway of exposure is through ingestion of food that is contaminated with bisphenol. Bisphenol can leach slowly into the food from things like the inside lining of cans, plastic containers, packages, disposable tableware, and especially through wear and tear. [19] This means that even simply using or eating food that came in contact with product made from bisphenol could result in exposure. [20] Moreover, studies have found bisphenol residue in foods and beverages even when using containers that claimed to be "bisphenol free".[20] [21] [22]
Dermal (Skin) Exposure
[edit]Bisphenol can also enter the body through the skin via absorption, and can result in skin irritation or allergic reactions in some individuals.[19][23] One well-known example is the handling of store receipts, which are often coated with bisphenol, and can transfer from the skin of the hands to the mouth, resulting in ingestion of the chemical.[24] Workers who handle receipts regularly, like cashiers and retail employees, may face higher dermal exposure.[19] [25]
Inhalation Exposure
[edit]Due to its volatility, bisphenol do not easily evaporate into the air at room temperature according to their chemical properties.[25][23] However, bisphenol can attach to dust an small particles in the environment and be inhaled. Usually, inhalation exposure of bisphenol are more common under industrial and manufacture settings. [26][18][27]
References
[edit]- ^ Fiege H, Voges HW, Hamamoto T, Umemura S, Iwata T, Miki H, et al. (2002). "Phenol Derivatives". Phenol Derivatives. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_313. ISBN 978-3-527-30673-2..
- ^ a b c Almeida S, Raposo A, Almeida-González M, Carrascosa C (November 2018). "Bisphenol A: Food Exposure and Impact on Human Health". Comprehensive Reviews in Food Science and Food Safety. 17 (6): 1503–1517. doi:10.1111/1541-4337.12388. PMID 33350146.
- ^ Vandenberg LN, Maffini MV, Sonnenschein C, Rubin BS, Soto AM (February 2009). "Bisphenol-A and the great divide: a review of controversies in the field of endocrine disruption". Endocrine Reviews. 30 (1): 75–95. doi:10.1210/er.2008-0021. PMC 2647705. PMID 19074586.
- ^ Bilbrey J (11 August 2014). "BPA-Free Plastic Containers May Be Just as Hazardous". Scientific American. Retrieved 8 August 2015.
- ^ a b Akash MS, Alshahrani S, Alzahrani A (2023). "Toxicological evaluation of bisphenol analogues: Preventive measures and therapeutic interventions". Environmental Toxicology and Pharmacology. 83: 103545. doi:10.1016/j.etap.2023.103545 (inactive 17 November 2025).
{{cite journal}}: CS1 maint: DOI inactive as of November 2025 (link) CS1 maint: article number as page number (link) - ^ a b Heinälä M, Ylinen K, Tuomi T, Santonen T, Porras SP (2017). "Assessment of occupational exposure to bisphenol A in five different production companies in Finland". Annals of Work Exposures and Health. 61 (1): 44–55. doi:10.1093/annweh/wxw006. PMID 28395312.
- ^ Hines CJ, Jackson MV, Deddens JA, Clark JC, Ye X, Christianson AL, et al. (2017-03-01). "Urinary Bisphenol A (BPA) Concentrations among Workers in Industries that Manufacture and Use BPA in the USA". Annals of Work Exposures and Health. 61 (2): 164–182. doi:10.1093/annweh/wxw021. ISSN 2398-7308. PMC 5577557. PMID 28395354.
- ^ Ribeiro E, Ladeira C, Viegas S (2017). "Occupational Exposure to Bisphenol A (BPA): A Reality That Still Needs to Be Unveiled". Toxics. 5 (3): 22. doi:10.3390/toxics5030022.
- ^ "Bisphenols". European Chemicals Agency. Retrieved 2025-11-10.
- ^ Rifa RA, Lavado R (2024). "Unveiling the next generation of bisphenol analogs and their impact on human health using in vitro methods". Emerging Contaminants. 10 (2): 100296. doi:10.1016/j.emcon.2023.100296.
{{cite journal}}: CS1 maint: article number as page number (link) - ^ Pelch KE, Wignall JA, Goldstone AE, Ross PK, Blain RB, Shapiro AJ, et al. (2017). NTP Research Report on Biological Activity of Bisphenol A (BPA) Structural Analogues and Functional Alternatives: Research Report 4. NTP Research Reports. Research Triangle Park (NC): National Toxicology Program. PMID 31944638.
- ^ Fishburn JL, Larson HL, Nguyen A, Welch CJ, Moore T, Penn A, et al. (2024-03-01). "Bisphenol F affects neurodevelopmental gene expression, mushroom body development, and behavior in Drosophila melanogaster". Neurotoxicology and Teratology. 102 107331. Bibcode:2024NTxT..10207331F. doi:10.1016/j.ntt.2024.107331. PMID 38301979.
- ^ Pivnenko K, Pedersen GA, Eriksson E, Astrup TF (October 2015). "Bisphenol A and its structural analogues in household waste paper" (PDF). Waste Management. 44: 39–47. Bibcode:2015WaMan..44...39P. doi:10.1016/j.wasman.2015.07.017. PMID 26194879. S2CID 217938141.
- ^ Im J, Löffler FE (2016). "Fate of Bisphenol a in Terrestrial and Aquatic Environments". Environmental Science & Technology. 50 (16): 8403–8416. doi:10.1021/acs.est.6b00877. OSTI 1470902. PMID 27401879.
- ^ See Bisphenol A#Environmental effects for extensive discussion
- ^ "Occupational Exposure to Bisphenol A (BPA) in U.S. Manufacturing Companies". NIOSH Science Blog. Centers for Disease Control and Prevention (CDC). 2017-01-05. Retrieved 2025-11-10.
- ^ "Occupational Exposure to BPA". CBIA. 2017-01-18. Retrieved 2025-11-10.
- ^ a b Hines CJ, Jackson MV, Deddens JA, Clark JC, Ye X, Christianson AL, et al. (2017). "Urinary Bisphenol A concentrations among workers". Annals of Work Exposures and Health. 61 (2): 164–182. doi:10.1093/annweh/wxw021.
- ^ a b c Lee SS, Ryu HY, Ahn KS, Lee S, Lee J, Lee JW, et al. (2022). "Toxicological profile of bisphenol F". Journal of Toxicology and Environmental Health Part A. 85 (4): 163–174. doi:10.1080/15287394.2021.1997843.
- ^ a b Benhamada M, Bouzid D, Boyron O, Taam M (2016). "Aging of polycarbonate and BPA release". European Food Research and Technology. 242 (2): 227–232.
- ^ Agarwal A, Gandhi S, Tripathi AD, Iammarino M, Homroy S (2022). "Bisphenol A migration from microwaveable cups". International Journal of Food Science & Technology. 57: 7477–7485.
- ^ Ali M, Jaghbir M, Salam M, Al-Kadamany G, Damsees R, Al-Rawashdeh N (2019). "Testing baby bottles for BPA". Environmental Monitoring and Assessment. 191 (1): 1–11.
- ^ a b National Institute for Occupational Safety and Health (2011). NIOSH Skin Notation Profiles: Bisphenol A (BPA) (Report). U.S. Department of Health and Human Services.
- ^ Ehrlich S, Calafat AM, Humblet O, Smith T, Hauser R (2014). "Handling of thermal receipts as a source of exposure to bisphenol A". JAMA. 311 (8): 859–860. doi:10.1001/jama.2013.283735.
- ^ a b National Institute of Environmental Health Sciences. "Bisphenol A (BPA)".
- ^ European Chemicals Agency (2023). Scientific report for evaluation of limit values for Bisphenol A at the workplace (Report).
- ^ Konieczna A, Rutkowska A, Rachoń D (2015). "Health risk of exposure to bisphenol A". Roczniki Państwowego Zakładu Higieny. 66 (1).
Further reading
[edit]- For additional examples and alternate names, see: Alger M (2017). Polymer Science Dictionary. Springer. p. 77. ISBN 978-94-024-0893-5.
